
So this is one, this one version of hydrogen. So this isn't actually what an atom looks like, but it's a very simply view that helps you get started. I'm going to draw that one electron somewhere outside the nucleus and I'm going to use the oversimplified Bohr model. 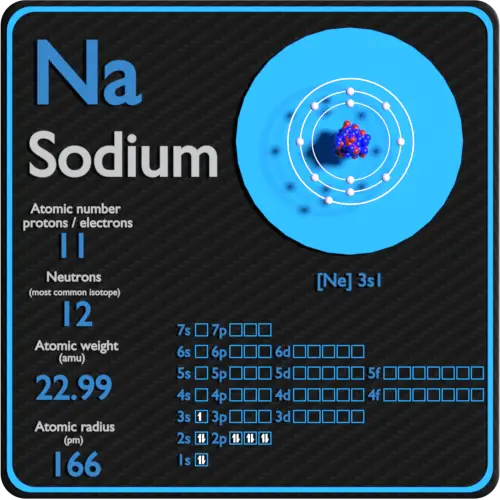
So there's my one proton in the nucleus, and we're talking about a neutral hydrogen atom, so there's one electron. 
We know the atomic number of hydrogen is one, so there's one proton in the nucleus. So let's go ahead and draw an atom of hydrogen. In a neutral atom, the number of protons is equal to the number of electrons, because in a neutral atom there's no overall charge and the positive charges of the protons completely balance with the negative charges of the electrons. So it's right here, so there's one proton in the nucleus of a hydrogen atom. So for hydrogen, hydrogen's atomic number is one. So we're going to talk about hydrogen in this video. And you can find the atomic number on the periodic table. So the atomic number is symbolized by Z and it refers to the number of protons in a nucleus. There are ways it can happen, but they are not the normal course of events. So you do not usually have one isotope changing into another isotope of the same element. So, it is not possible to give you a general answer other than to say that the isotopes of an element form in one or more of the above ways, but each isotope of an element may or may not form in a similar way as other isotopes.Įxcept as listed above, an atom that is not radioactive never changes its number of neutrons. There are a few other ways, but they are not all that significant.Įach isotope (more properly called a nuclide) has its own way(s) in which it is formed. Being changed from one type of atom to another by high energy nuclear reactions, such as having a neutron slam into the nucleus at an extremely high velocity. With the exception of the hydrogen and helium that were formed shortly after the Big Bang event, elements mainly form in the following ways:Ĥ.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
